New book on Prion Diseases from Cold Spring Harbor Laboratory Press
07/27/2017
Diseases such as Creutzfeldt-Jakob disease and kuru develop when PrP proteins form prions by misfolding, clumping together, and spreading from cell to cell. Over the past decade, a number of proteins have been reported to possess the characteristics of PrP prions. A growing collection of disorders has been found to be caused by prions; some of these prion diseases include such devastating illnesses as Alzheimer's and Parkinson's and possibly type 2 diabetes. Written and edited by experts in the field, Prion Diseases covers the progress that has recently been made in our understanding of the pathophysiology of prion diseases, as well as ongoing efforts to develop effective therapeutics. The contributors discuss how proteins such as tau, islet amyloid polypeptide, and α-synuclein adopt alternative shapes that lead them to aggregate, resulting in cellular degeneration. Therapies for human and animal diseases caused by prions are also covered. This volume is therefore useful for all biomedical scientists and physicians wishing to understand and treat this expanding group of devastating disorders. Please visit our website to access free samples and for further information on this title. | 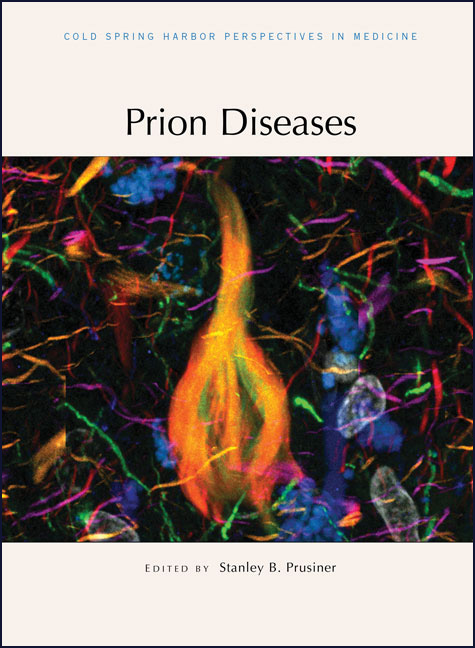
IMAGE: Confocal microscopy image reveals an intracellular neurofibrillary tangle (center; yellow-orange) and surrounding neurophil threads (magenta, orange, red and green) found in a human Alzheimer's disease brain postmortem. These hallmark pathological structures are composed of misfolded tau proteins double labeled by a monoclonal antibody and a fluorescent small-molecule amyloid-binding probe. Unaffected cells (white) and intracellular stress granules (blue) composed of lipofuscin are visualized adjacent to the pathologic tau deposits. This image was produced from a confocal "z-stack" consisting of numerous optical sections through 30 u.m of tissue and subsequently flattened using image-processing software. CREDIT:Carlo Condello, University of California, San Francisco
|
 Cold Spring Harbor Laboratory Press Bookstore
Cold Spring Harbor Laboratory Press Bookstore

